Role of dexmedetomidine for the prevention and treatment of delirium in intensive care unit patients. Dexmedetomidine vs midazolam or propofol for sedation during prolonged mechanical ventilation: two randomized controlled trials. Jakob SM, et al Dexmedetomidine for Long-Term Sedation Investigators.Dexmedetomidine: A Review of Its Use for Sedation in the Intensive Care Setting. Dexmedetomidine for the management of awake fibreoptic intubation. Dexmedetomidine: an adjuvant making large inroads into clinical practice. Among patients with agitated delirium receiving mechanical ventilation in the Intensive Care Unit, the addition of dexmedetomidine to standard care compared with standard care alone resulted in more ventilator-free hours at 7 days. 
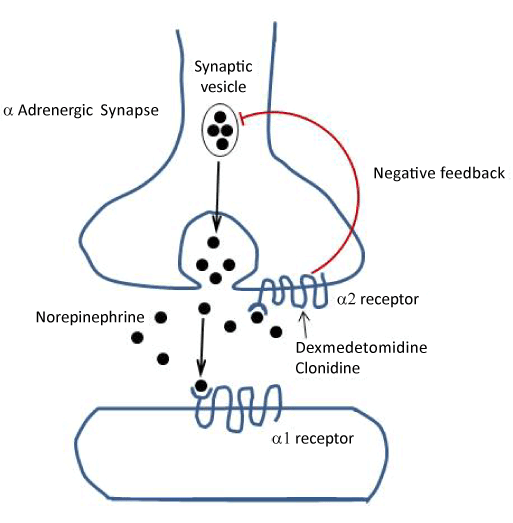
few patients enrolled despite screening >20,000 patients, which may limit external validity.Clinicians could use dexmedetomidine as a rescue agent in the control arm.Sedation received prior to randomisation was not well characterised.Patients in the dexmedetomidine group were ventilated for longer prior to enrolment in the study (median times were 63 hours vs 43.5 hours).underpowered (did not reach the calculated n=96 sample size), yet still found a difference.Multiple secondary outcomes, some favoured dexmedetomidine and none favoured the control arm.increase in median ventilator free hours at 7 days in the dexmedetomidine group: 144.8 vs.Intervention: Dexmedetomidine started at 0.5mcg/kg/hr and then titrated to rates between 0 and 1.5mcg/kg/hr to achieve RASS 0 or a physician-prescribed goal clinical-directed option for bolus of 1.0mcg/kg over 20 minutes.n =71 adult ICU patients who needed to remain mechanically ventilated because their degree of agitation was considered so severe as to make lessening their sedation and extubation unsafe.Dose utilised up to twice the maximum 0.7mcg/kg/hr dosing approved by FDA in US.One author (not a lead) disclosed personal payment from Orion.Trial designed and controlled by Orion Pharma (patent holders of dexmedetomidine), including statistical analyses and manuscript preparation (listed as co-author).MV duration) tested as part of a superiority trial using intention-to-treat analysis Non-inferiority trial powered (90%) to test the hypothesis that dexmedetomide is no more than 15% worse than propofol (PRODEX) or midazolam (MIDEX) for time to reach sedation target using per-protocol analysis.Significantly more bradycardia (14% vs 5%).Shorter MV duration: extubated 2 days sooner.Incidence of hypotension / bradycardia (13% each).Duration of MV: extubated sooner with dexmed however required NIV post.Exclusion criteria: narrow, reasonable (pregnancy, shock, pre-existing bradycardia etc).Dexmedetomidine (0.2 – 1.4mcg/kg/hr) versus propofol (PRODEX) or midazolam (MIDEX).DB MC RCT mixed non-inferiority and superiority trial.Jakob SM, et al Dexmedetomidine for Long-Term Sedation Investigators. Reduced ICU delirium and MV duration in the dexmedetomidine group.Comparable in achieving sedation targets in MV patients.Dexmedetomidine compared with midazolam.only licensed for use in mechanically ventilated patients for 24 hours.ventilator dysynchrony due to discomfort.loading dose 1mcg/kg over 10 minutes (optional).suppression of stress response to surgery and other noxious stimuli.decreases CBF, CNS VO2, and mild decrease in ICP.decreases circulating and cerebral catecholamines and CNS glutamate.peripheral nerve mechanisms also implicated.posterior horns of the spinal cord where the modulation of pain impulses is mediated by the noradrenergic bulbar/spinal pathway.minimal respiratory depression (RR, PaCO2, SpO2, and higher PF ratios).produces a patient who is sedated but can be easily roused with minimal stimulation.sedation is induced by inhibition of noradrenergic activity via activation of alpha-2 receptors at the locus coeruleus.reduces sympathetic activity and agitation, causing a state resembling the non-REM phase of sleep without impairing cognitive function.binds all 3 subtypes of alpha2 receptor (A, B, C). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
